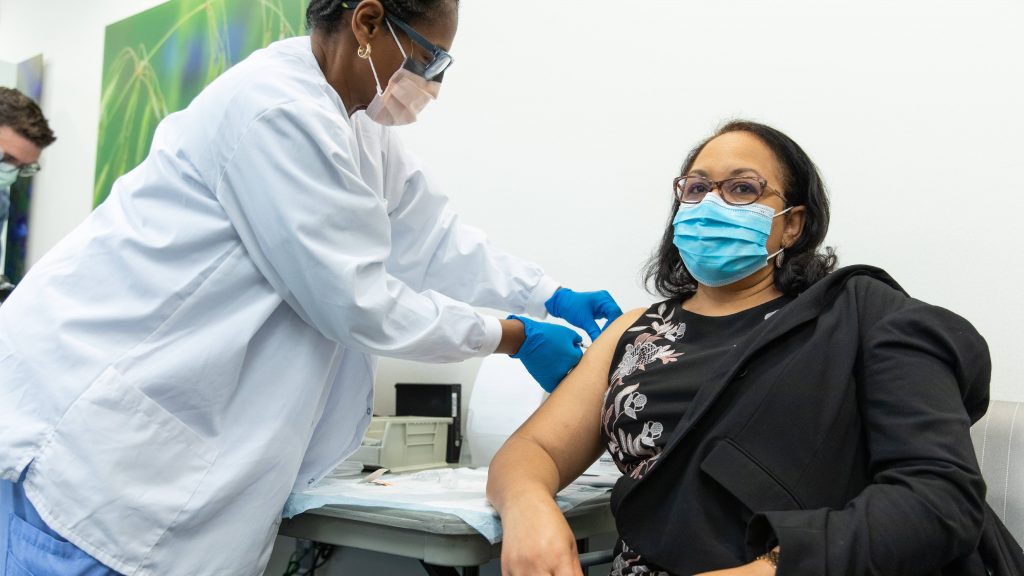
President Joe Biden says the U.S. soon will be able to vaccinate 1.5 million people daily for COVID-19, and he expects any person who wants to be vaccinated will be able to do so by the spring.
Dr. Melanie Swift, co-chair of Mayo Clinic's COVID-19 Vaccine Allocation and Distribution Work Group, says the Pfizer/BioNTech and Moderna vaccines are approximately 95% effective in preventing COVID-19.
"That's an incredibly good number. I mean, when we look at our annual flu vaccine, the effectiveness of that is only about 50% to 60%. So this is incredibly good news that we have vaccines that are this effective," says Dr. Swift.
Watch: Dr. Melanie Swift discusses COVID-19 vaccine safety and efficacy
Journalists: Broadcast-quality sound bites with Dr. Swift are available in the downloads. Please courtesy "Melanie Swift, M.D./COVID-19 Vaccine Allocation and Distribution/Mayo Clinic."
She says the vaccines, which are administered in two doses spaced 21 or 28 days apart, are especially safe because they do not contain the live virus that causes COVID-19. Instead, they are messenger RNA vaccines that trigger an immune response that teaches the body's cells to fight off the virus by producing antibodies.
"The immune response begins with the first dose. And then, with the second dose, it intensifies. And it will take about two weeks after that second dose before the immune response has fully developed. So, from two weeks after your second dose is probably a good time to expect you'll have your maximum protection," says Dr. Swift.
No COVID-19 vaccines are available yet for children under 16. But several companies have begun enrolling children in COVID-19 vaccine clinical trials.
"The Moderna vaccine is only authorized for use in ages 18 and up, and the Pfizer vaccine is authorized in ages 16 and up. So we don't yet have a vaccine that is actually authorized for anybody under 16 years old," says Dr. Swift. "There will be more trials in March recruiting and enrolling subjects who are ages 5 to 11. So we will ultimately have a vaccine for the pediatric population, but we don't have one of those yet."
If you've had an immediate allergic reaction to other vaccines or injectable medications, ask your health care provider if you should be vaccinated for COVID-19. If you've ever had an immediate allergic reaction to any ingredient in one of the COVID-19 vaccines, such as polysorbate, the Centers for Disease Control and Prevention recommends not getting that specific vaccine.
_______________________________
For the safety of its patients, staff and visitors, Mayo Clinic has strict masking policies in place. Anyone shown without a mask was either recorded prior to COVID-19 or recorded in a nonpatient care area where social distancing and other safety protocols were followed.
Information in this post was accurate at the time of its posting. Due to the fluid nature of the COVID-19 pandemic, scientific understanding, along with guidelines and recommendations, may have changed since the original publication date.
For more information and all your COVID-19 coverage, go to the Mayo Clinic News Network and mayoclinic.org.
Learn more about: Tracking COVID-19 and COVID-19 trends