-
Featured News
Asthma Discovery May Lead to New Therapeutic Approach
Embargoed: Do not release before 2:00 p.m. U.S. Eastern Time, April 22, 2015
Mayo Clinic researchers and collaborators have discovered a key cellular mechanism that contributes to bronchoconstriction and inflammation in asthma. Their studies may lead to a novel, effective asthma therapy for patients. The findings appear in the journal Science Translational Medicine.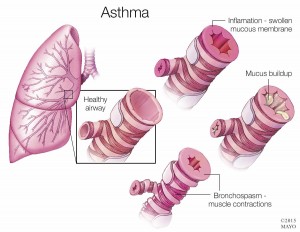
The team of researchers from Mayo Clinic, Cardiff University in Wales, King’s College London, the University of Manchester and the Open University in the United Kingdom, as well as the University of California San Francisco, showed that elevated extracellular calcium as well as other internal chemicals released in asthma can activate a “calcium-sensing receptor” (CaSR) on airway cells. The researchers found that CaSR expression is increased in asthma, and contributes to enhanced bronchoconstriction and inflammation that is typically seen in asthma. Importantly, they found that the effects of CaSR can be reversed by CaSR antagonists, also called calcilytics.
Media Contact: Bob Nellis at Mayo Clinic Public Affairs: 507-284-5005 or newsbureau@mayo.edu
Asthma is a growing concern worldwide, with over 235 million people diagnosed according to the World Health Organization, and the largest chronic disease among children. WHO says over 80 percent of asthma-related deaths occur in the under-developed countries.
“Our findings point to an entirely novel pathophysiological mechanism in asthma. As our studies proceed, if we can show that calcilytics can target multiple cell types and mechanisms that contribute to asthma or even other lung diseases, then we may have an exciting new therapeutic target,” says Y.S. Prakash, M.D., Ph.D., Mayo Clinic anesthesiologist, physiologist, and co-senior author of the study. “Calcilytics are small molecules and so they could be readily delivered by inhalation, thus limiting potential systemic side effects.”
Co-authors include Alecia Stewart, Venkatachalem Sathish, Rodney Britt Jr., Ph.D., Michael Thompson, Michelle Freeman, Bharathi Aravamudan, Ph.D., Hirohito Kita, M.D., and Christina Pabelick, M.D., of Mayo Clinic; Polina Yarova, Ph.D., Sarah Brennan, Ph.D., Martin Schepelmann, Ph.D., Thomas Davies, Sun Yung, Ph.D., Paul Kemp, Ph.D., Alexander Lowe, Zakky Cholisoh, Emma Kidd, Ph.D., William Ford, Ph.D., Kenneth Broadly, Ph.D., and Daniela Ricardi, Ph.D., of Cardiff University; Katja Rietdorf, Ph.D., The Open University; Wenhan Chang, Ph.D., University of California-SF; Mohamed Bin Khayat and Donald Ward, Ph.D., University of Manchester; and Christopher Corrigan, Ph.D., and Jeremy Ward, Ph.D., King’s College London.
Funding for the research came from Asthma UK, the Cardiff Partnership Fund, the Marie Curie Initial Training Network, the Biotechnology and Biological Sciences Research Council, and the U. S. National Institutes of Health. Drs. Ricardi, Kemp,