-
Mayo Clinic and United Therapeutics to increase lungs for transplantation in new building dedicated to medical discovery, innovation

JACKSONVILLE, Fla. — Mayo Clinic in Florida's new Discovery and Innovation Building opened on Thursday, Aug. 22. Here, pioneering technology will increase the number of lungs available for transplant. The 75,000-square-foot building also will house an innovative Life Sciences Incubator that connects entrepreneurs with resources to bring medical solutions to market.
Lungs are among the most fragile and difficult organs to transplant. They are susceptible to injuries and infections, with only about 20% of donor lungs in the U.S. meeting the standard for transplant. On average, about 300 people die every year waiting for a lung transplant. About 1,400 people are waiting for a lung transplant, and many more could benefit from a transplant if more viable lungs were available, according to the United Network for Organ Sharing, the organization that oversees the nation's organ transplant system.
To address the shortage of suitable lungs, Mayo Clinic and United Therapeutics (NASDAQ: UTHR) announced a collaboration in 2015 that combines the use of United Therapeutics' ex-vivo lung perfusion technology and Mayo Clinic's medical expertise. With ex-vivo lung perfusion (EVLP), donor lungs are flushed and ventilated while being monitored in isolation. This process allows transplant programs to re-evaluate lungs that would otherwise have been discarded.
As part of the agreement, Mayo Clinic leased land at its Florida location to Lung Biotechnology PBC, a wholly owned subsidiary of United Therapeutics, to construct the building, and equip and operate the center. The lungs will be perfused by Lung Bioengineering, a subsidiary of Lung Biotechnology PBC. Mayo Clinic will provide physician oversight and work with organ procurement organizations to coordinate the procurement and delivery of lungs. The lungs will be made available to patients at Mayo Clinic and other transplant centers. Mayo Clinic and United Therapeutics also will collaborate on the manufacturing of cell-based therapies for regenerative medicine research, an innovative area of medicine that could heal damaged tissue and organs.
"We are excited to work with United Therapeutics in this important and lifesaving collaboration that ultimately will increase the number of lungs for transplant patients," says Kent Thielen, M.D., CEO of Mayo Clinic in Florida. "Mayo Clinic is firmly committed to using and developing the latest medical advances and innovations that will have tremendous benefits not only for our patients, but other patients at institutions throughout the U.S."
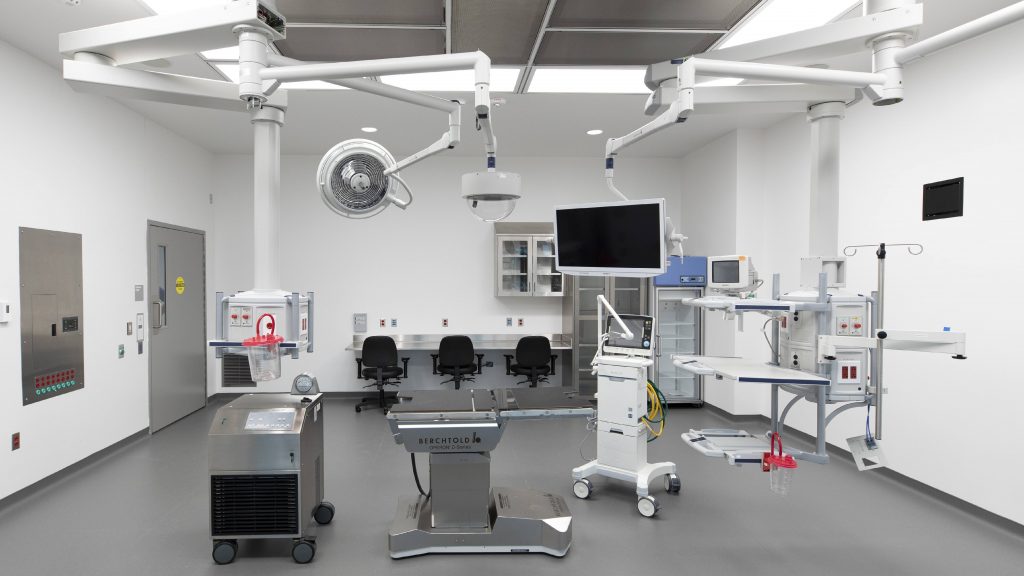
The building includes three ex-vivo lung perfusion surgical suites on the first floor and collaborative space for conducting cell-based therapy production on the second floor. The Mayo Clinic Life Sciences Incubator is on the third floor.
"We are honored to work with Mayo Clinic to expand the supply of transplantable lungs," said Martine Rothblatt, chairman and CEO of United Therapeutics. "Hundreds of additional transplantable lungs have been made available through EVLP technology, and we have more than doubled the number of hours that a lung can be outside of a body before being safely transplanted. In addition we hope to prevent the need for some lung transplants and to treat bronchopulmonary dysplasia with new biologic medicines we will manufacture in the new building."
Mayo Clinic has one of the nation's largest and most experienced transplant practices, with locations in Arizona, Florida and Minnesota. More than 200 physicians in transplant medicine and surgery at these three locations perform about 1,800 organ transplants per year and have a long track record of excellent outcomes.
Mayo Clinic established its lung transplant program in Florida in 2001. Since then, Mayo has performed 648 lung transplants and nine heart-lung transplants. The three-year survival rate at Mayo Clinic in Florida exceeds the national average by 9%. The patient survival rate at Mayo is 82% versus 73% nationally.
Mayo Clinic Life Sciences Incubator
To further accelerate advances in health care, the Mayo Clinic Life Sciences Incubator will bring together a community of innovators from across Northeast Florida, providing a hub for entrepreneurs to explore ideas; develop new health care and life science products and services; and create new, thriving companies.
The Mayo-led incubator programs will help:
- Inventors and entrepreneurs who wish to bring new ideas, products or technologies to market
- Educators that would like to partner with the business community
- Students that seek experiential learning opportunities
- Companies that want to access new ideas, products and talent to grow their business portfolios
- Economic development agencies that are eager to attract new business opportunities to the region
- Investors searching for new capital growth opportunities
"The Mayo Clinic Life Sciences Incubator will provide entrepreneurs in the community with a dedicated facility and structured programming that links local startups to an array of regional business, science and investment resources — a partnership of significant and numerous regional assets," says Charles Bruce, M.D., chief innovation officer, Mayo Clinic in Florida. "In turn, a lasting business environment will thrive in the Jacksonville area that spurs new solutions for patients around the world." Dr. Bruce is medical director for the incubator.
The Life Sciences Incubator received a competitive $750,000 federal grant in July from the U.S. Economic Development Administration. This i6 Challenge grant is designed to fuel high-growth entrepreneurship and increase access to capital for communities across the U.S. This grant is awarded to just 26 organizations. Mayo's incubator will use the funds to collaborate with a dozen regional partners, contributing $12 million to the economy and creating 120 jobs in the Jacksonville area.
Dr. Bruce, an accomplished innovator who has helped create seven startup companies and holds 21 patents for medical inventions, says the award is significant not only for the amount, but also for the recognition.
"This award is a clear sign of Mayo Clinic's commitment to the region, our efforts to grow the economy, and attracting top talent in the life sciences field," Dr. Bruce says. "Mayo Clinic has earned the confidence and pledged collaboration from the research, business and economic development communities throughout Northeast Florida, as well as the backing of local and state government agencies."
Accelerated growth
Over the past four years, Mayo Clinic in Florida has flourished with major construction and new staff to serve a fast-growing patient population, especially those who require complex medical care. During this time, Mayo Clinic has invested more than $600 million in construction projects and added more than 1,000 new staff members. Two months ago, Mayo Clinic in Florida announced another new major construction project: a $233 million integrated oncology facility that includes proton beam therapy. The 140,000-square-foot facility, which is expected to be completed in late 2023, will be located next to the Mangurian Building.
Mayo Clinic in Florida has more than 6,000 staff members and contributes nearly $2 billion annually to the Florida economy.
###
About United Therapeutics
United Therapeutics Corporation focuses on the strength of a balanced, value-creating biotechnology model. The corporation is confident in its future thanks to its fundamental attributes, namely its obsession with quality and innovation, the power of its brands, its entrepreneurial culture and its bioinformatics leadership. The corporation also believes that its determination to be responsible citizens — having a positive impact on patients, the environment and society — will sustain its success in the long term.
Through our wholly owned subsidiary, Lung Biotechnology PBC, United Therapeutics is focused on addressing the acute national shortage of transplantable lungs and other organs with a variety of technologies that either delay the need for such organs or expand the supply. Lung Biotechnology is the first public benefit corporation subsidiary of a public biotechnology or pharmaceutical company. [uthr-g]
About Mayo Clinic
Mayo Clinic is a nonprofit organization committed to innovation in clinical practice, education and research, and providing compassion, expertise and answers to everyone who needs healing. Visit the Mayo Clinic News Network for additional Mayo Clinic news and An Inside Look at Mayo Clinic for more information about Mayo.
Forward-looking statements
Statements included in this press release that are not historical in nature are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, among others, United Therapeutics' and Mayo's expectations regarding the collaboration between United Therapeutics and Mayo Clinic, and the ability of this collaboration to result in an increase in the available supply of lungs for transplant. These forward-looking statements are subject to certain risks and uncertainties, such as those described in United Therapeutics' periodic reports filed with the Securities and Exchange Commission (SEC) that could cause actual results to differ materially from anticipated results. Consequently, such forward-looking statements are qualified by the cautionary statements, cautionary language and risk factors set forth in United Therapeutics' periodic reports and documents filed with the SEC, including the most recent Annual Report on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. United Therapeutics claims the protection of the safe harbor contained in the Private Securities Litigation Reform Act of 1995 for forward-looking statements. This information is provided as of August 22, 2019, and neither United Therapeutics nor Mayo Clinic assumes any obligation to update or revise the information contained in this press release whether as a result of new information, future events or any other reason.
Media contacts:
- Kevin Punsky, Mayo Clinic Public Affairs, 904-953-0746, punsky.kevin@mayo.edu
- Tia Ford, Mayo Clinic Public Affairs, 904-953-1419, ford.tia@mayo.edu
- James Edgemond, United Therapeutics, 301-608-9292, jedgemond@unither.com