DEAR MAYO CLINIC: I've heard several drugs mentioned as possible treatments for COVID-19. What are they, and how do they work?
ANSWER: Since the COVID-19 pandemic began, finding an effective drug to combat COVID-19 has been on the minds of many. While many medications are being tested to treat COVID-19, only one has been approved by the Food and Drug Administration (FDA).
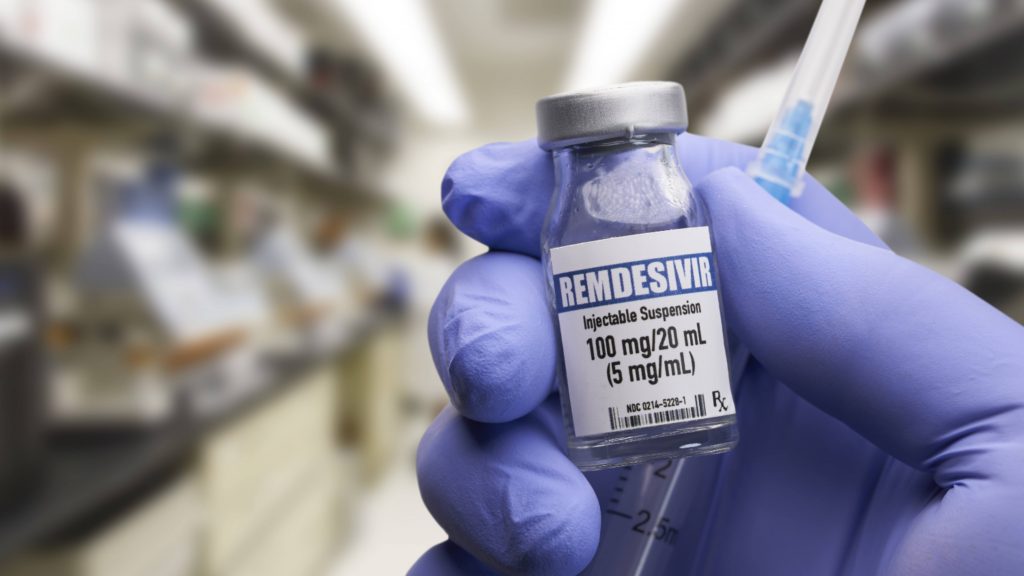
The FDA has approved remdesivir (Veklury) to treat COVID-19 in adults and children who are age 12 and older. Remdesivir is an IV antiviral drug that can be prescribed for people who are hospitalized with COVID-19.
The FDA has granted an emergency use authorization for baricitinib (Olumiant), a rheumatoid arthritis drug, to treat COVID-19 in some cases. Baricitinib is a pill that seems to work against COVID-19 by reducing inflammation. The FDA states baricitinib can be used in people who are hospitalized with COVID-19 who are on mechanical ventilators or need supplemental oxygen.
Researchers are studying other potential treatments for COVID-19, including:
Antiviral drugs
In addition to remdesivir, other antiviral drugs being tested include favipiravir and merimepodib. Studies have found that the combination of lopinavir and ritonavir isn't effective. An experimental oral drug known as monupiravir has been reported to be effective in reducing the risk of hospitalization and death in non-hospitalized patients at high risk for COVID-19. Recently, the drug's manufacturer, Merck, indicated plans to seek emergency use authorization for the drug.
Dexamethasone
Dexamethasone, a corticosteroid, is one type of anti-inflammatory drug that researchers are studying to treat or prevent organ dysfunction and lung injury from inflammation. Studies have found that it reduces the risk of death by about 30% for people on ventilators and by about 20% for people who needed supplemental oxygen.
The National Institutes of Health has recommended this drug for people hospitalized with COVID-19 who are on mechanical ventilators or need supplemental oxygen. Other corticosteroids, such as prednisone, methylprednisolone or hydrocortisone, can be used if dexamethasone isn't available. Dexamethasone and other corticosteroids may be harmful if given for less severe COVID-19 infection.
In some cases, tocilizumab or baricitinib can be given with dexamethasone in hospitalized people who are on mechanical ventilation or need supplemental oxygen. Remdesivir may be given with dexamethasone in hospitalized people who need supplemental oxygen or who are on mechanical ventilation.
Anti-inflammatory therapy
Researchers study many anti-inflammatory drugs to treat or prevent dysfunction of several organs and lung injury from infection-associated inflammation.
Immune-based therapy
The FDA granted emergency use authorization in 2020 for a type of immune-based therapy called convalescent plasma to treat COVID-19. Convalescent plasma is blood donated by people who've recovered from COVID-19. Convalescent plasma was being studied as to its effectiveness in treating some hospitalized people ill with COVID-19 who are early in their illness or have weakened immune systems. Though recently it was reported to not be as effective as first thought, studies are still ongoing.
Researchers also study other immune-based therapies, including mesenchymal stem cells and monoclonal antibodies. Monoclonal antibodies are proteins created in a lab that can help the immune system fight off viruses. Several monoclonal antibody medications are available. These include sotrovimab and a combination of two antibodies called casirivimab and imdevimab. These drugs are used to treat mild to moderate COVID-19 in people who have a higher risk of developing serious illness due to COVID-19. Treatment consists of a single IV infusion given in an outpatient setting. To be most effective, these medications need to be given soon after COVID-19 symptoms start and prior to hospitalization.
Drugs being studied that have uncertain effectiveness
Researchers are studying amlodipine and losartan. But it's not yet known how effective these drugs may be in treating or preventing COVID-19. Ivermectin and famotidine aren't likely to help treat COVID-19.
Hydroxychloroquine and chloroquine
These malaria drugs were authorized for emergency use by the FDA during the COVID-19 pandemic. However, the FDA withdrew that authorization when data analysis showed that the drugs are not effective for treating COVID-19. They also can cause serious heart problems.
Drugs to prevent COVID-19.
Researchers are studying drugs to prevent COVID-19 before and after exposure to the virus.
In summary, it's not known if any of these treatments will prove effective against COVID-19. Medical studies will need to be completed to determine their effectiveness against COVID-19.
Don't try these medications without a prescription and your health care provider's approval, even if you've heard that they may have promise. These drugs can have serious side effects. They're reserved for people who are seriously ill and under a health care provider's care. — Dr. Daniel DeSimone, Infectious Diseases, Mayo Clinic, Rochester, Minnesota
____________________________________________
Information in this post was accurate at the time of its posting. Due to the fluid nature of the COVID-19 pandemic, scientific understanding, along with guidelines and recommendations, may have changed since the original publication date.
For more information and all your COVID-19 coverage, go to the Mayo Clinic News Network and mayoclinic.org.
Learn more about tracking COVID-19 and COVID-19 trends.