Children 12–15 are now eligible to receive Pfizer's COVID-19 vaccine, after the U.S. Food and Drug Administration granted emergency use approval.
And there is other COVID-19 news.
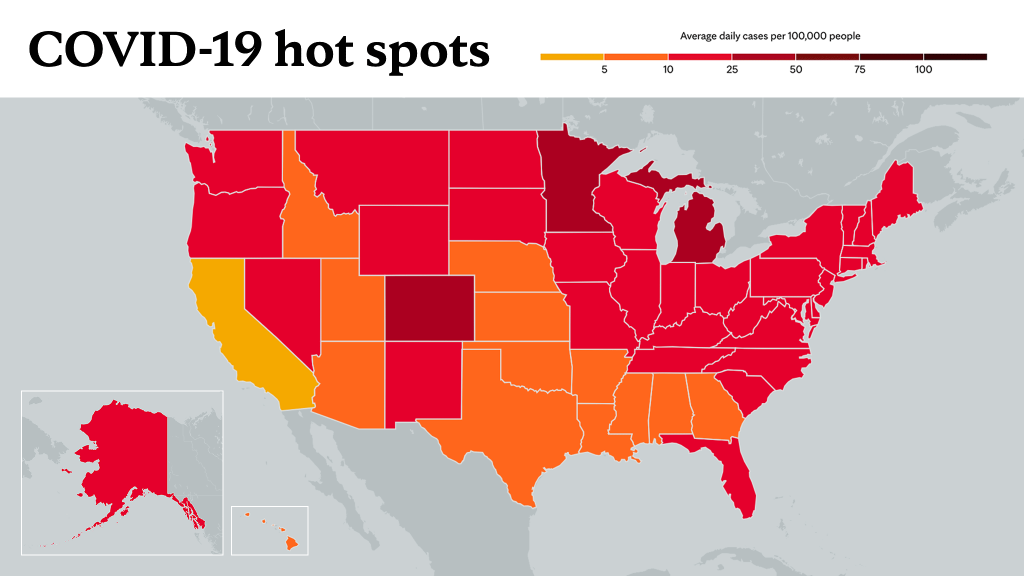
"This is the first week we have not had an increase in COVID-19 cases in any U.S. state, and in 33 of those states, there was a decrease last week," says Dr. Gregory Poland, an infectious diseases expert and head of Mayo Clinic's Vaccine Research Group. "This has not happened on any consistent basis for every U.S. state since the start of this pandemic."
In this Mayo Clinic Q&A podcast, Dr. Poland discusses the real-world effectiveness of the COVID-19 vaccines and the approval process for these vaccines, and he answers a number of listener questions.
Watch: Dr. Poland talks about real world effectiveness of the COVID-19 vaccines.
Read the full transcript.
____________________________________________
For the safety of its patients, staff and visitors, Mayo Clinic has strict masking policies in place. Anyone shown without a mask was either recorded prior to COVID-19 or recorded in a nonpatient care area where social distancing and other safety protocols were followed.
Information in this post was accurate at the time of its posting. Due to the fluid nature of the COVID-19 pandemic, scientific understanding, along with guidelines and recommendations, may have changed since the original publication date.
Research disclosures for Dr. Gregory Poland.
For more information and all your COVID-19 coverage, go to the Mayo Clinic News Network and mayoclinic.org.
Learn more about tracking COVID-19 and COVID-19 trends.