The Centers for Disease Control and Prevention (CDC) has updated its recommendations for the Pfizer COVID-19 vaccine to expand the use of a single booster dose to 12- to 15-year-olds. The Food and Drug Administration amended its emergency use authorization earlier this week.
"Everyone at age 12 and up is approved for a booster after they've completed their initial series," says Dr. Melanie Swift, co-chair of Mayo Clinic's COVID-19 Vaccine Allocation and Distribution Work Group.
Watch: Dr. Melanie Swift clears up confusion about COVID-19 boosters.
Journalists: Broadcast-quality sound bites with Dr. Swift are available in the downloads at the end of the post. Please courtesy: "Melanie Swift, M.D./COVID-19 Vaccine Allocation and Distribution/Mayo Clinic."
What constitutes an initial series depends on the COVID-19 vaccine you were given. If it was Johnson & Johnson, it's one shot. If it was an mRNA vaccine, which is Moderna or Pfizer, an initial series is two shots. And if you are immunocompromised and were vaccinated with Moderna or Pfizer, it's three shots.
"To keep it clear, just remember that it's your initial series, however many doses that is, and one booster for now," says Dr. Swift. "At least that is in the United States right now. And that's, with everything COVID, always subject to change."
"The timing of the booster depends upon the brand of your initial series. If you got Johnson & Johnson, it's two months. If you got Pfizer initially, it's five months. If you got Moderna initially, it's six months. Now that might change in the future. But that's the current guidance based upon the available data," she says.
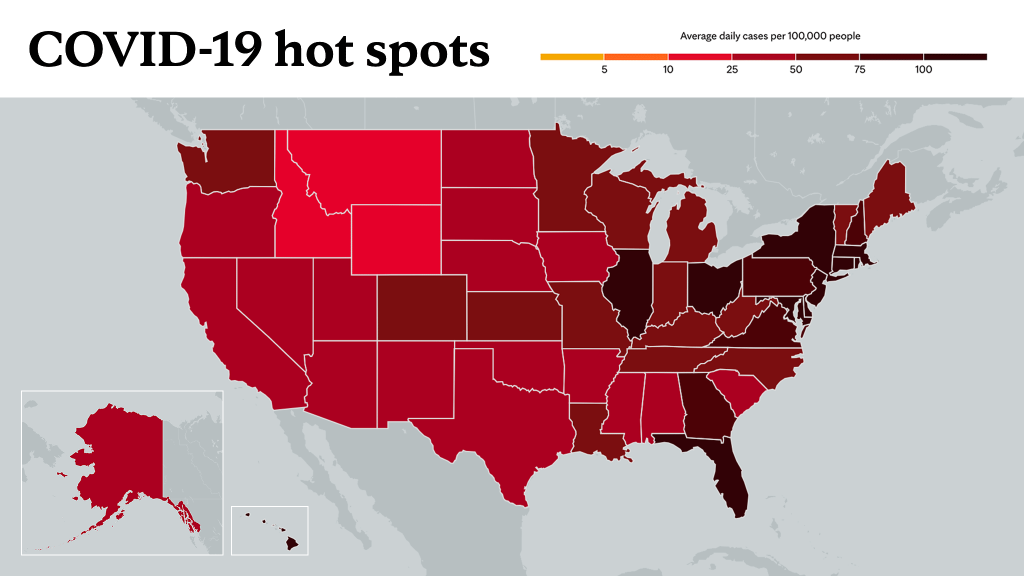
This comes at a time when the omicron variant is spreading rapidly across the nation. The U.S. recently hit a new pandemic high of over 300,000 average new daily cases of COVID-19. And COVID-19 hospitalizations are on the rise, as well.
"We know that one of the things about omicron is that it makes your vaccines less effective. The vaccination protection with two doses of an mRNA vaccine against omicron is down in the 40-something percent range — so not great. But with a booster, that protection goes up over 75%," says Dr. Swift. "We know that in addition to the fact that your immunity just wanes over time, omicron, in particular, is more susceptible to your immune response after the third or booster dose. So we are really encouraging people to get that booster dose."
For the safety of its patients, staff and visitors, Mayo Clinic has strict masking policies in place. Anyone shown without a mask was either recorded prior to COVID-19 or recorded in a nonpatient care area where social distancing and other safety protocols were followed.
Information in this post was accurate at the time of its posting. Due to the fluid nature of the COVID-19 pandemic, scientific understanding, along with guidelines and recommendations, may have changed since the original publication date.
For more information and all your COVID-19 coverage, go to the Mayo Clinic News Network and mayoclinic.org.
Learn more about tracking COVID-19 and COVID-19 trends.