-
Mayo scientists reverse fibrosis in preclinical studies
ROCHESTER, Minn. — In cell and mouse models, Mayo Clinic researchers and collaborators have identified a way to slow and reverse the process of uncontrolled internal scarring, called fibrosis. This disease process has few effective therapies, no cure and can be fatal when it occurs in organs such as the liver (cirrhosis) or lungs (pulmonary fibrosis). The findings appear Wednesday, Oct. 30, in Science Translational Medicine.
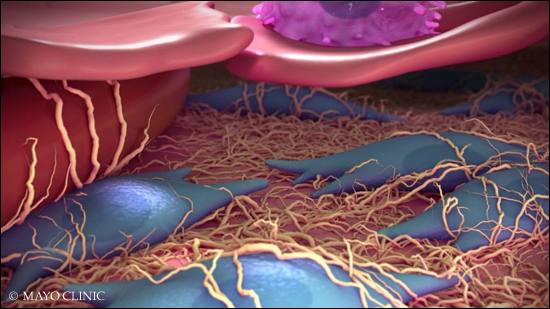
In the past, researchers identified two proteins that regulate gene action and provide "instructions" for fibrosis, called yes-associated protein (YAP) and transcriptional co-activator with PDZ-binding motif (TAZ). However, YAP and TAZ are involved in too many other beneficial actions and can't be universally blocked. So the scientists, led by Daniel Tschumperlin, Ph.D., a Mayo Clinic researcher, focused on cells that form the scar tissue, called fibroblasts. After an injury, these cells migrate to the damaged area and produce collagen to re-form the scaffolding around cells and repair tissue. In fibrosis, fibroblasts release collagen in amounts that interfere with an organ's function.
"We started this work almost five years ago with seed funding from Mayo's Center for Biomedical Discovery because fibrosis is an important and poorly understood disease process," explains Dr. Tschumperlin. "We had identified the transcriptional regulatory proteins YAP and TAZ as important to the progression of fibrosis, but there were no viable approaches to target this pathway for therapy."
The publication reports on a new target: a dopamine receptor. The authors found that the receptor is unique to lung and liver fibroblasts. Stimulating it blocks YAP and TAZ, reducing and reversing the growth and scar-forming abilities of fibroblasts. Stimulating the dopamine receptor in fibroblasts appeared to switch them from a matrix-depositing state that supports fibrosis to a matrix-degrading state that supports resolution or reversal of fibrosis. In lung and liver fibrosis models, the approach reversed the fibrotic process in these organs.

"Dopamine is mostly studied in disorders of the central nervous system, and we were surprised to find dopamine receptors expressed in fibroblasts," says Dr. Tschumperlin. "We also found that the lung may be able to produce dopamine locally, and this function appears to be compromised in individuals with pulmonary fibrosis. Thus, the pathway we are targeting might be part of a normal response that would limit fibrosis, but is somehow missing in patients."
This result, the researchers say, opens up a new treatment concept for fibrotic disease conditions. They hope to build on the decades of work that already has gone into developing dopamine receptor-targeting drugs in the brain to develop new dopamine-related therapies for fibrosis. "In addition to following up on regulation of dopamine signaling in the lung, we are actively developing novel molecules to target the dopamine receptor, as we think there is substantial promise in trying to translate this approach to human diseases," says Dr. Tschumperlin.
Other authors of the paper include collaborators from the University of Pittsburgh, Massachusetts General Hospital/Harvard Medical School, and Boston University School of Medicine. In addition to Dr. Tschumperlin, other Mayo authors are Andrew Haak, Ph.D.; Enis Kostallari, Ph.D.; Delphine Sicard, Ph.D.; Giovanni Ligresti, Ph.D.; Kyoung Choi, Ph.D.; Nunzia Caporarello, Ph.D.; Dakota Jones, a student in Mayo Clinic Graduate School of Biomedical Sciences; Qi Tan, Ph.D.; Jeffrey Meridew; Ana Diaz Espinosa; Aja Aravamudhan, Ph.D.; Jessica Maiers, Ph.D.; Rodney Britt Jr., Ph.D.; Anja Roden, M.D.; Christina Pabelick, M.D.; Y.S. Prakash, M.D., Ph.D.; and Vijay Shah, M.D.
Funding for this work was provided by federal and nongovernmental sources, as listed in the publication, and support was provided by Mayo Clinic's Center for Clinical and Translational Science.
The first author, Dr. Haak, and Dr. Tschumperlin disclose that they are co-inventors of a patent application based on some of the findings in the publication.
###
About Mayo Clinic
Mayo Clinic is a nonprofit organization committed to innovation in clinical practice, education and research, and providing compassion, expertise and answers to everyone who needs healing. Visit the Mayo Clinic News Network for additional Mayo Clinic news and An Inside Look at Mayo Clinic for more information about Mayo.
Media contact:
- Sara Tiner, Mayo Clinic Public Affairs, 507-284-5005, newsbureau@mayo.edu