-
Cancer
New Immunotherapy Approved for Metastatic Bladder Cancer
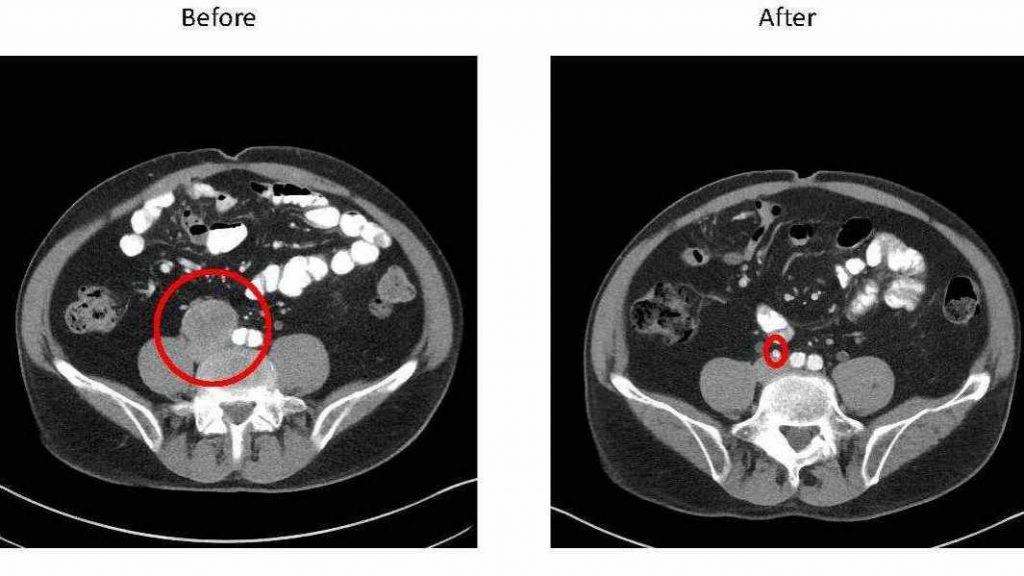 Patients with metastatic bladder cancer have few treatment options after failure of chemotherapy until now.
Patients with metastatic bladder cancer have few treatment options after failure of chemotherapy until now.
In early May, the U.S. Food and Drug Administration (FDA) approved the immunological drug Atezolizumab to treat metastatic bladder cancer. The decision came following results of an international, multisite phase II clinical study published in the March 4 edition of the Lancet. Mayo Clinic’s Florida campus was one of the largest sites involved in the study.
Dr. Richard Joseph, a Mayo Clinic oncologist involved in the study, says the drug works by helping the patient’s own immune system fight cancer.
“Bladder cancer is the ninth most common cancer, causing 165,000 deaths worldwide annually,” says Dr. Joseph, whose research is part of the Cancer Immunology and Immunotherapy Program within the Mayo Clinic Cancer Center.
“Until now, treatment options for patients with metastatic bladder cancer were limited. Chemotherapy has been the only option for these patients, but many people are unable to tolerate the full course of chemotherapy, due to its toxicity. New approaches, such as immunotherapy medications like this, are desperately needed.”
Watch: Dr. Joseph explains the study, how the new drug works and treatment options.
Journalists: Broadcast-quality sound bites with Dr. Joseph are in the downloads.
Of the 310 patients involved in the study, 15–20 percent had significant and lasting response, says Dr. Joseph, which is part of what led to the FDA's fast approval. "The response rates lead us to believe these patients will have very long and durable remissions," he adds.
Tumors can avoid immune system surveillance by expressing genes that turn off the immune system and prevent it from recognizing them. Atezolizumab inhibits one such gene present on the surface of bladder cancer tumor cells, thereby allowing the immune system to recognize and attack the tumor.
Immunotherapy drugs have also recently been approved for melanoma, kidney cancer and lung cancer.







