
Allergan textured breast implants and tissue expanders are being removed from the U.S. market, due to the risk of breast implant-associated anaplastic large cell lymphoma. That's according to a recent statement from the maker and the Food and Drug Administration (FDA).
"BIOCELL®saline-filled and silicone-filled textured breast implants and tissue expanders will no longer be distributed or sold in any market where they are currently available. Effective immediately, health care providers should no longer implant new BIOCELL® textured breast implants and tissue expanders and unused products should be returned to Allergan."
"We support Allergan and the FDA’s decision to remove the BIOCELL® textured breast implants from the market to allow further investigation of its link to breast implant-associated anaplastic large cell lymphoma (BIA-ALCL)," says Dr. Minh-Doan Nguyen, a Mayo Clinic plastic surgeon.
"Mayo Clinic has been proactive in addressing this issue and is currently in compliance with the FDA recommendations," says Dr. Nguyen. "We have been using smooth tissue expanders since March 2019, when they became available, and we notified patients from the past 20 years who have had textured implants and have seen them, as needed."
In addition, Dr. Nguyen says Mayo agrees with current recommendations that preventative removal of textured implants in patients who do not have any symptoms of BIA-ALCL is not necessary. However patients are encouraged to discuss their concerns with their plastic surgeon. The most common symptom for BIA-ALCL is formation of a delayed fluid pocket or seroma around the implant. It can also present as a new breast mass or skin lesion.
_________________________________________________________________
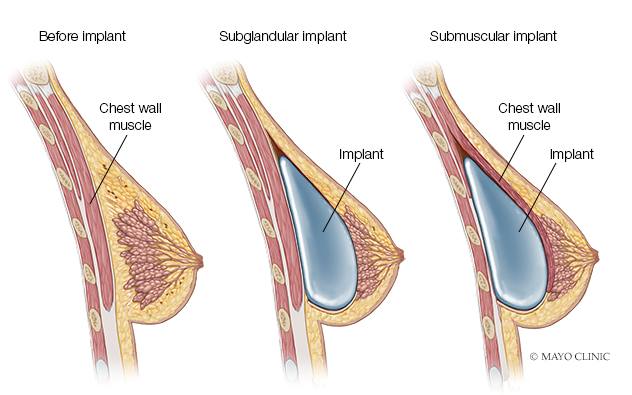
Breast Augmentation Overview
Breast augmentation — also known as augmentation mammoplasty — is surgery to increase breast size. It involves placing breast implants under breast tissue or chest muscles.
For some women, breast augmentation is a way to feel more confident. For others, it's part of rebuilding the breast for various conditions.
If you're considering breast augmentation, talk to a plastic surgeon. Make sure you understand what surgery involves, including possible risks, complications and follow-up care.
Why it's done
Breast augmentation might help you:
- Enhance your appearance if you think your breasts are too small or that one is smaller than the other
- Adjust for a reduction in the size of your breasts after pregnancy
- Correct uneven breasts after breast surgery for other conditions
- Improve your self-confidence
Discuss your goals with your surgeon so that you can be realistic about what breast augmentation can do for you.
Risks
Breast augmentation poses various risks, including:
- Scar tissue that distorts the shape of the breast implant (capsular contracture)
- Breast pain
- Infection
- Changes in nipple and breast sensation
- Implant leakage or rupture
Correcting these complications might require more surgery, either to remove or replace the implants.
The FDA has identified a possible association between breast implants and the development of breast implant-associated anaplastic large cell lymphoma (ALCL), a rare cancer of the immune system. The FDA believes that women with breast implants might have a very low but increased risk of developing ALCL. However, further research is needed to fully understand the relationship between ALCL and breast implants.
Before you decide to have surgery, consider the following:
- Breast implants won't prevent your breasts from sagging. To correct sagging breasts, you might need a breast lift in addition to breast augmentation.
- Breast implants aren't guaranteed to last a lifetime. The average life span of an implant is 10 years. Implant rupture is a possibility. Also, your breasts will continue to age, and factors such as weight gain or weight loss might change the way your breasts look. These issues will likely lead to more surgery.
- Mammograms might be more complicated. If you have breast implants, in addition to routine mammograms, you'll require additional, specialized views.
- Breast implants might hamper breastfeeding. Some women are able to successfully breastfeed after breast augmentation. For others, however, breastfeeding is a challenge.
- Insurance does not cover breast implants. Unless it's medically necessary — such as after a mastectomy — breast augmentation isn't covered by insurance. Be prepared to handle the expenses, including related surgeries or future imaging tests.
- You might need additional surgery after breast implant removal. If you decide to have your implants removed, you might need a breast lift or other corrective surgery to help restore your breasts' appearance.
- You might need an MRI scan. The FDA recommends routine monitoring with MRI after three years. However, recent studies show that there's little data to support routine screening unless you have symptoms.
Sometimes, breast augmentation is done during local anesthesia — you're awake and your breast area is numbed. Often, though, breast augmentation is done during general anesthesia, in which you're asleep for the surgery.
Learn more about this procedure.








