-
American Medical Society for Sports Medicine position on regenerative medicine
The American Medical Society for Sports Medicine has released a position statement to guide responsible use of regenerative interventions when standard care doesn't provide relief. The statement recommends key scientific and ethical considerations when prescribing regenerative medicine procedures. The position statement published in the Clinical Journal of Sport Medicine emphasizes safety, scientific validation, ethics and patient education when integrating emerging regenerative therapies in the practice of sports medicine.
"In most cases, people heal musculoskeletal injuries with over-the-counter pain medication and conventional physical therapy. However, a subset of about 25% of people don't progress with standard care," says Shane Shapiro, M.D., a sports medicine physician and author of the" American Medical Society for Sports Medicine" position statement. "That's when we consider other scientifically based regenerative options. This new position statement outlines considerations for safely and responsibly applying regenerative medicine to clinical care." Dr. Shapiro is also the medical director of the Regenerative Medicine Therapeutic Suites at Mayo Clinic in Florida.
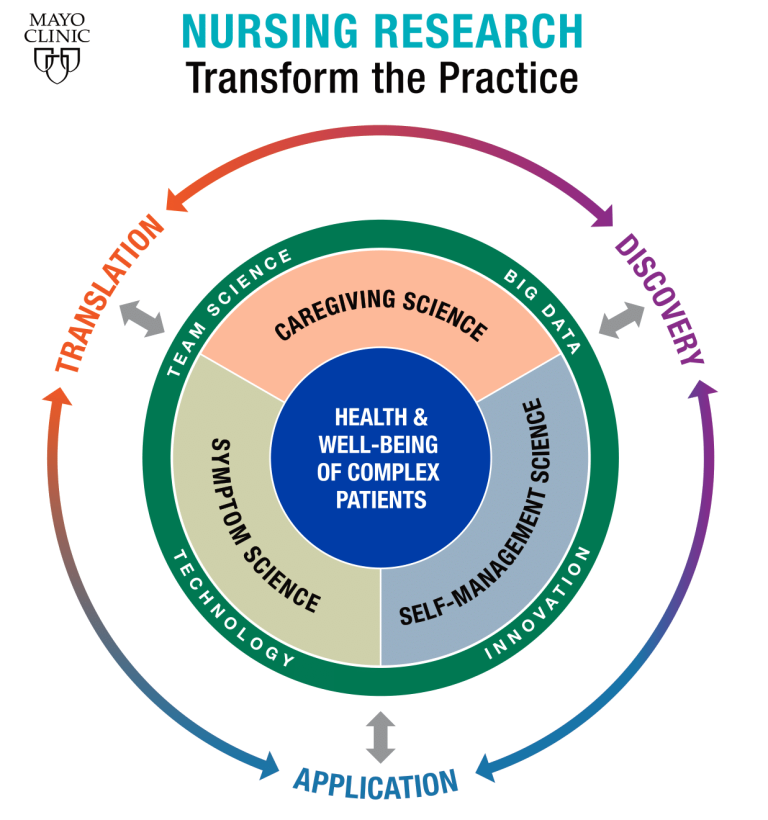
Regenerative medicine is a new field of practice that is shifting the focus of medicine to rebuilding health by repairing, replacing or restoring diseased cells, tissues and organs. Mayo Clinic's Center for Regenerative Medicine is at the vanguard of this movement. It supports research and practice that provide new therapeutic options in sports medicine.
The American Medical Society for Sports Medicine position statement offers six key considerations for sports medicine clinicians when using regenerative interventions:
Treatment gaps
Regenerative medicine becomes an alluring option for the many people within the treatment gaps for joint and musculoskeletal conditions. These are the people with torn ligaments or degenerative conditions like osteoarthritis who don't respond to over-the-counter pain relievers and physical therapy. Yet they are too young to be good candidates for surgery or joint replacements.
Practice considerations
After exhausting standard therapies, orthobiologics is a regenerative intervention that can bridge the treatment gap. Orthobiologics use biological agents with growth factors to activate healing. An example is platelet-rich plasma, which consists of platelets spun from a person's own blood that trigger healing.
"The least invasive, safest, cost-effective treatment with the strongest evidence should be implemented first. Only after these treatment options are exhausted should more invasive and novel treatments, such as regenerative medicine options, be considered," says Dr. Shapiro.
The position statement emphasizes that additional training may be needed for practitioners to safely deliver orthobiologic procedures to patients.
Evidence
Research has shown that platelet-rich plasma is safe, and it offers some benefits for knee osteoarthritis and certain tendinopathies. The position statement notes that beyond that, evidence does not support widespread routine use of orthobiologics for other conditions.
"Stronger evidence is required before other orthobiologic therapies can be routinely recommended," says Dr. Shapiro.
Informed consent
While emerging regenerative medicine therapies spur hope of new treatment, sports medicine practitioners warn against capitalizing on hype. Patients should be informed that regenerative interventions are not currently considered standard of care treatments. Information about the procedures should be presented in an honest and balanced manner regarding what is known and unknown, and what is scientifically proven versus what is not.
"Clinicians must provide understandable information to patients so they can make informed decisions about their care," says Dr. Shapiro.
Regulatory considerations
Regenerative medicine must comply with the regulatory guidelines of the Food and Drug Administration (FDA) in the U.S. and similar regulatory bodies around the world. Therapeutic use of human stem cells, tissues and tissue products must either be approved by the FDA or must align with FDA guidelines.
"The FDA recently clarified the need for premarket approval in order to use injectable perinatal cells and tissues in clinical practice, and to date, none has received the requisite biologic licenses," says Dr. Shapiro.
However, commonly used orthobiologics, such as platelet-rich plasma, often fall in the category of minimal manipulation of cells and are allowed in routine medical practice. A procedure is considered minimal manipulation if the cells are not combined with another substance and if the patient is being treated with his or her own cells. Clinicians should follow FDA guidelines when delivering procedures that are allowed under minimal manipulation of cellular products.
Standard terminology
Regenerative medicine is an emerging field with new terms that have yet to be standardized. To foster an understanding for patients and clinicians, the sports medicine position statement advocates for and defines a common regenerative medicine lexicon for sports medicine. It recommends those terms be used in future scientific publications and patient communications.
"Oftentimes, orthobiologics is described as a stem cell therapy, which is not the case. That is detrimental to the progress of regenerative medicine, and it is confusing to the patients and the public," says Dr. Shapiro.
The American Medical Society for Sports Medicine position paper concludes that the need for new scientifically validated procedures to address treatment gaps has generated interest in regenerative interventions. Sports medicine practitioners should advocate for advancing evidence, patient education and safety to bring promising new regenerative therapies to more sports medicine patients.
Zubin Master, Ph.D., a Mayo Clinic bioethicist, and Jonathan Finnoff, D.O., a Mayo Clinic supplemental researcher and medical director, U.S. Olympic & Paralympic Committee, also contributed to this sports medicine position statement.
###