-
Featured News
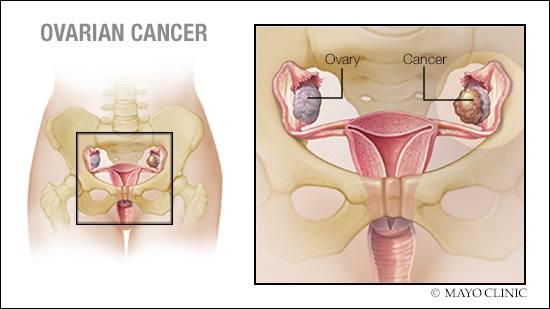
New surgical method for ovarian cancer lights up lesions
Mayo Clinic scientists are shining light on ovarian cancer.
A study published in 2022 in the Journal of Clinical Oncology explored the use of fluorescence-guided surgery to treat epithelial ovarian cancer, the most common type of ovarian cancer.
Fluorescence-guided surgery uses fluorescence imaging to light up cancerous tissue, giving surgeons a better view of tumors and cancer cells. This makes cancer removal more thorough and accurate.
"It was really exciting to be part of fluorescence-guided surgery," says Kristina Butler, M.D., a Mayo Clinic gynecologic oncologist and one of the study authors. "For ovarian cancer, we have evidence that removing as much of the tumor as possible at the time of surgery can improve a patient’s overall survival and help prevent the disease from progressing."
Before fluorescence-guided surgery, patients receive an injection of contrast dye that makes their cancer glow, explains Dr. Butler.
An estimated 19,710 people will receive an ovarian cancer diagnosis in the U.S. in 2023, according to the American Cancer Society. About 85% to 90% of those cancers will be epithelial ovarian cancer.
Detecting ovarian cancer in real-time with pafolacianine
The phase 3 study evaluated the safe and effective use of pafolacianine — a fluorescent drug that targets a folate receptor that may be overexpressed in ovarian cancer — with a type of fluorescence imaging called intraoperative near-infrared imaging (NIR).
The 109 study participants were people diagnosed with folate receptor-positive epithelial ovarian cancer or suspected of having the disease. Patients received one 60-minute infusion of pafolacianine one hour before surgery.
During the surgical procedure, all suspicious visible lesions (cancerous tissue) were documented for all patients using white light and palpation (examining by touch). Patients were then randomly assigned to be evaluated a second time using NIR fluorescence imaging or to proceed directly to surgery to remove the lesions.
In the second evaluation, the care team used NIR fluorescence imaging to identify additional lesions not visible under white light. Surgeons removed all visible lesions. Before completing surgery, they also reimaged the areas from which lesions were removed to ensure no visible lesions remained.
For both study groups, lesions were sent to a laboratory where pathologists evaluated the tissues for cancer. Pathologists weren’t told whether lesions had been identified using white light or NIR fluorescence imaging.
The lab results confirmed that NIR fluorescence imaging helped surgeons identify tumors that weren’t visible under white light.
"This technology can be used with newly diagnosed patients, patients who have already had chemotherapy and patients with recurrent ovarian cancer," says Dr. Butler. "It opens up opportunities that did not exist for patients before."
Future directions for fluorescence-guided surgery
The Food and Drug Administration approved the fluorescent drug used in this study for use in surgery. Researchers are now evaluating other drugs that may further enhance surgical outcomes and address side effects.
Dr. Butler says some drugs used in fluorescence-guided surgery can cause non-cancerous tissue, such as lymph nodes or necrotic (dead) tissue, to glow. This makes it difficult to determine which tissue is cancerous. And some drugs have a high rate of side effects, including nausea and vomiting.
Experts at Mayo Clinic have been at the forefront of the effort to bring new fluorescent drugs into clinical research.
"We're looking at other options that still provide enhanced outcomes of more tumor detection and removal," says Dr. Butler.
Dr. Butler and her team hope to next evaluate aminolevulinic acid hydrochloride, a drug approved for fluorescence-guided surgery of high-grade gliomas (a type of brain or spinal cord tumor) that accumulates in cancer cells. The team aims to learn how this drug’s ability to target cancerous tissue may be used in ovarian cancer surgery.
This article originally appeared on the blog of the Mayo Clinic Comprehensive Cancer Center.